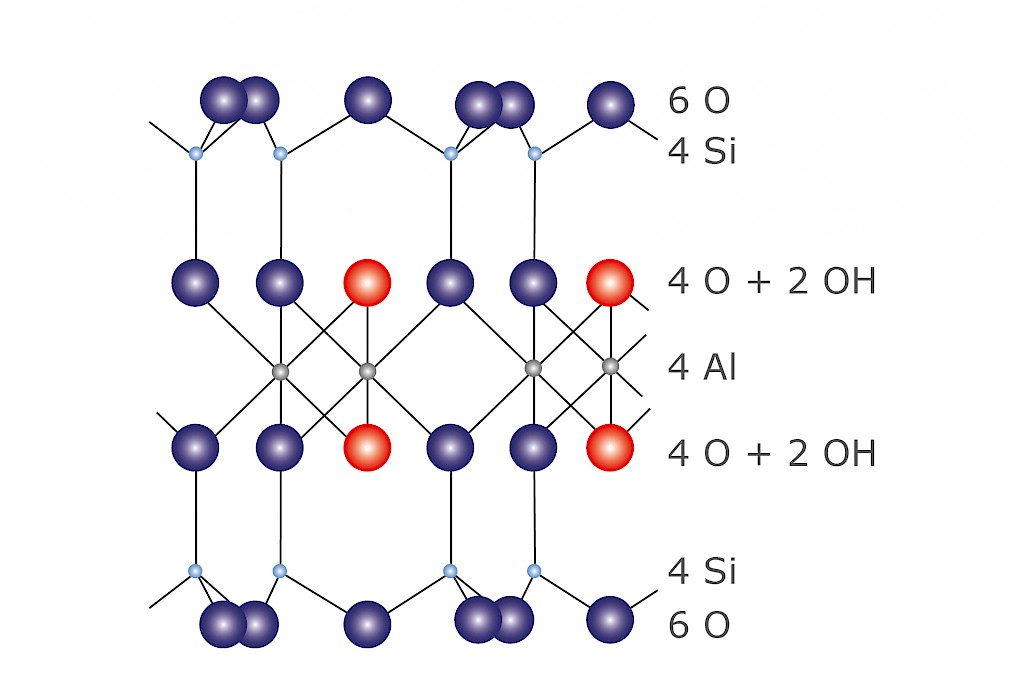
Surface spectroscopy and structural analysis of nanostructured multifunctional (Zn, Al) layered double hydroxides - Richetta - 2016 - Surface and Interface Analysis - Wiley Online Library
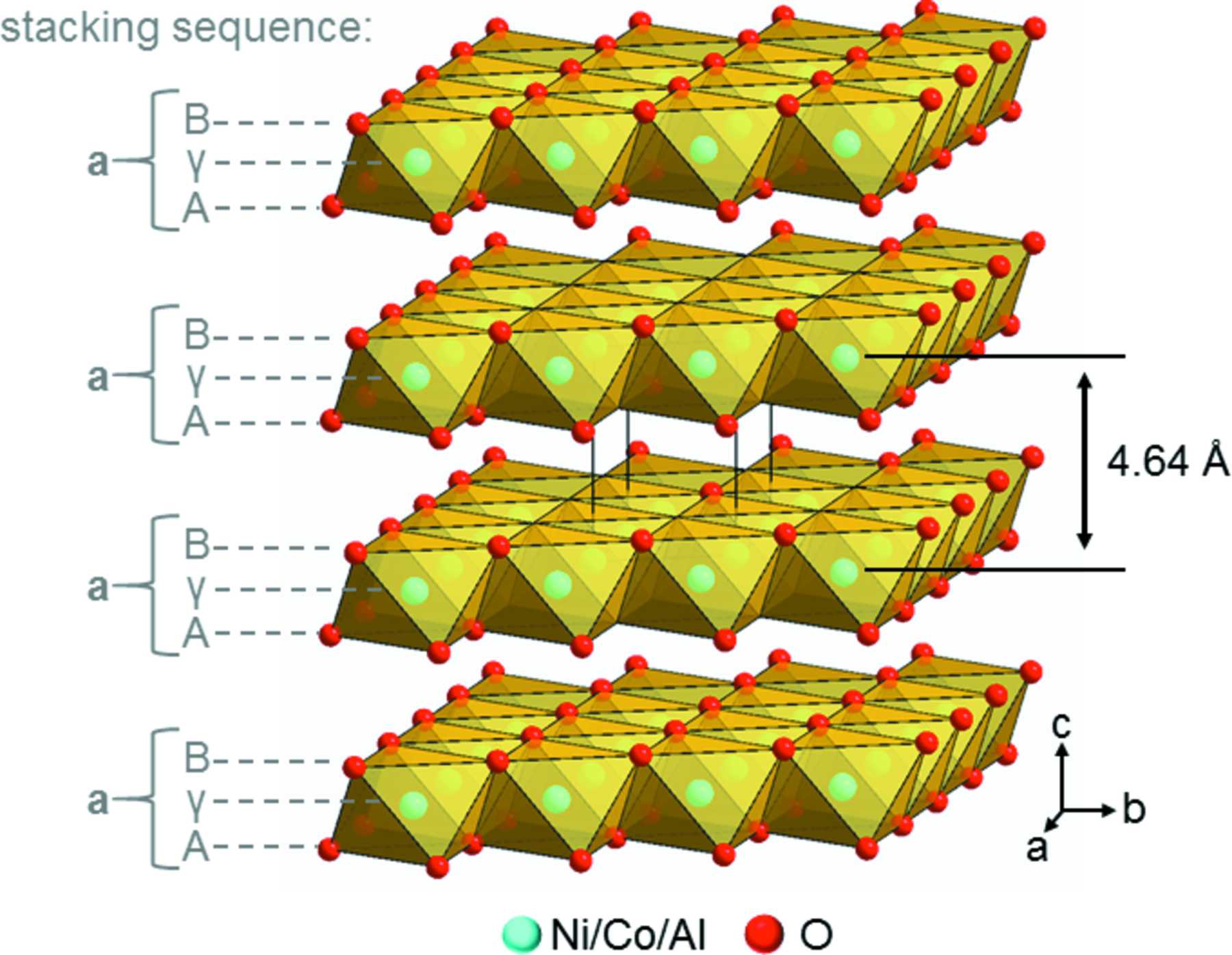
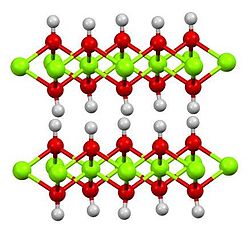
IUCr) A routine for the determination of the microstructure of stacking-faulted nickel cobalt aluminium hydroxide precursors for lithium nickel cobalt aluminium oxide battery materials

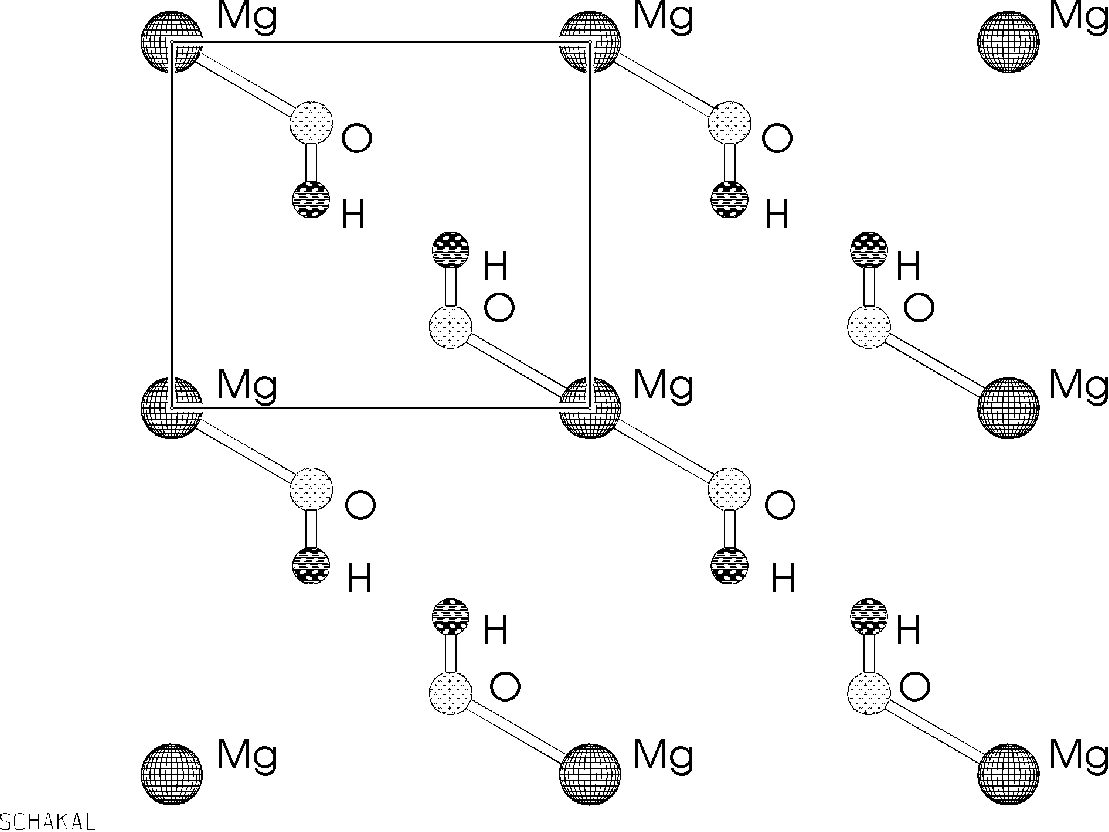
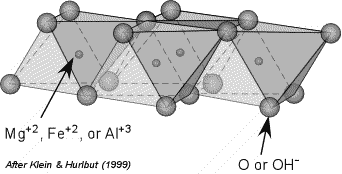
Figure 2 from Ionic adsorption on the brucite (0001) surface: A periodic electrostatic embedded cluster method study. | Semantic Scholar

Structure of Hydrated Gibbsite and Brucite Edge Surfaces: DFT Results and Further Development of the ClayFF Classical Force Field with Metal–O–H Angle Bending Terms | The Journal of Physical Chemistry C
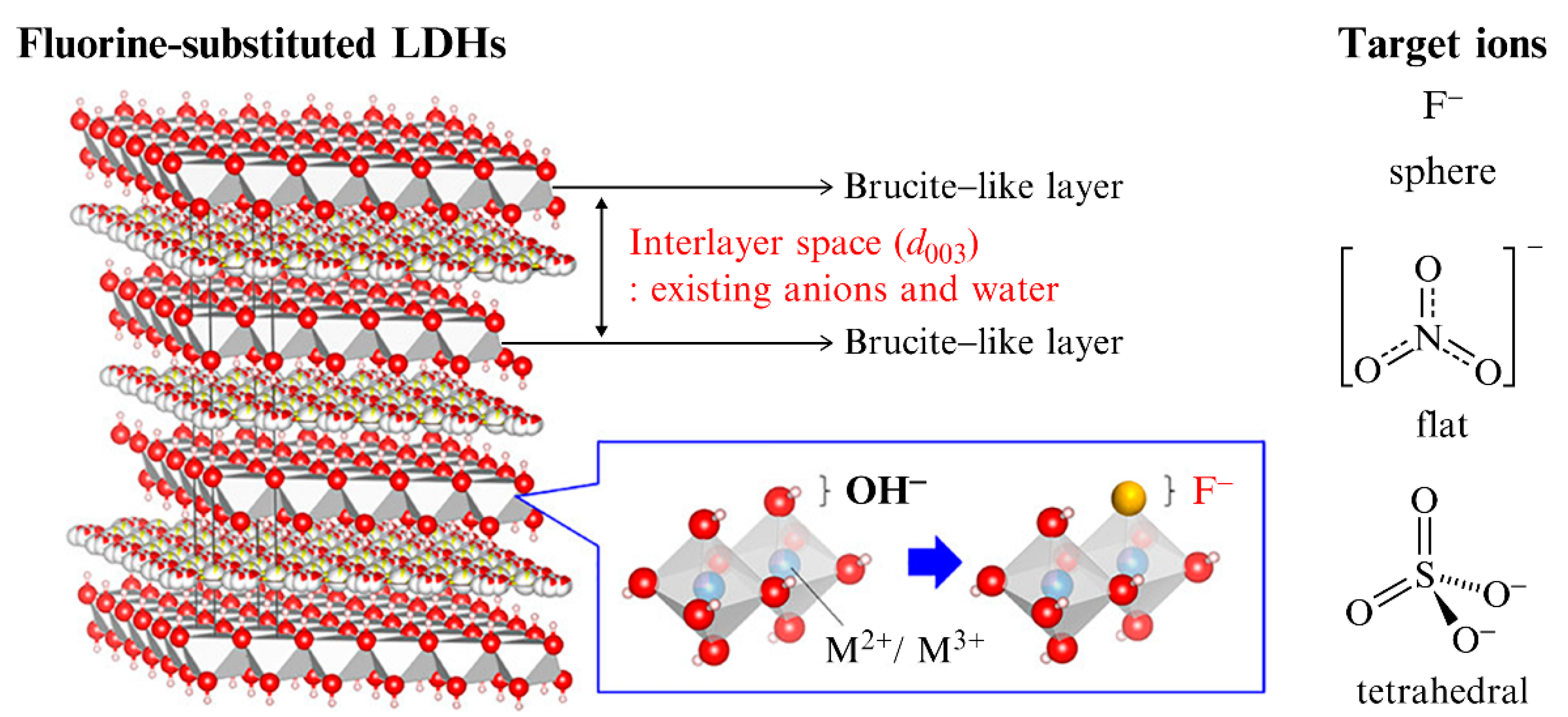
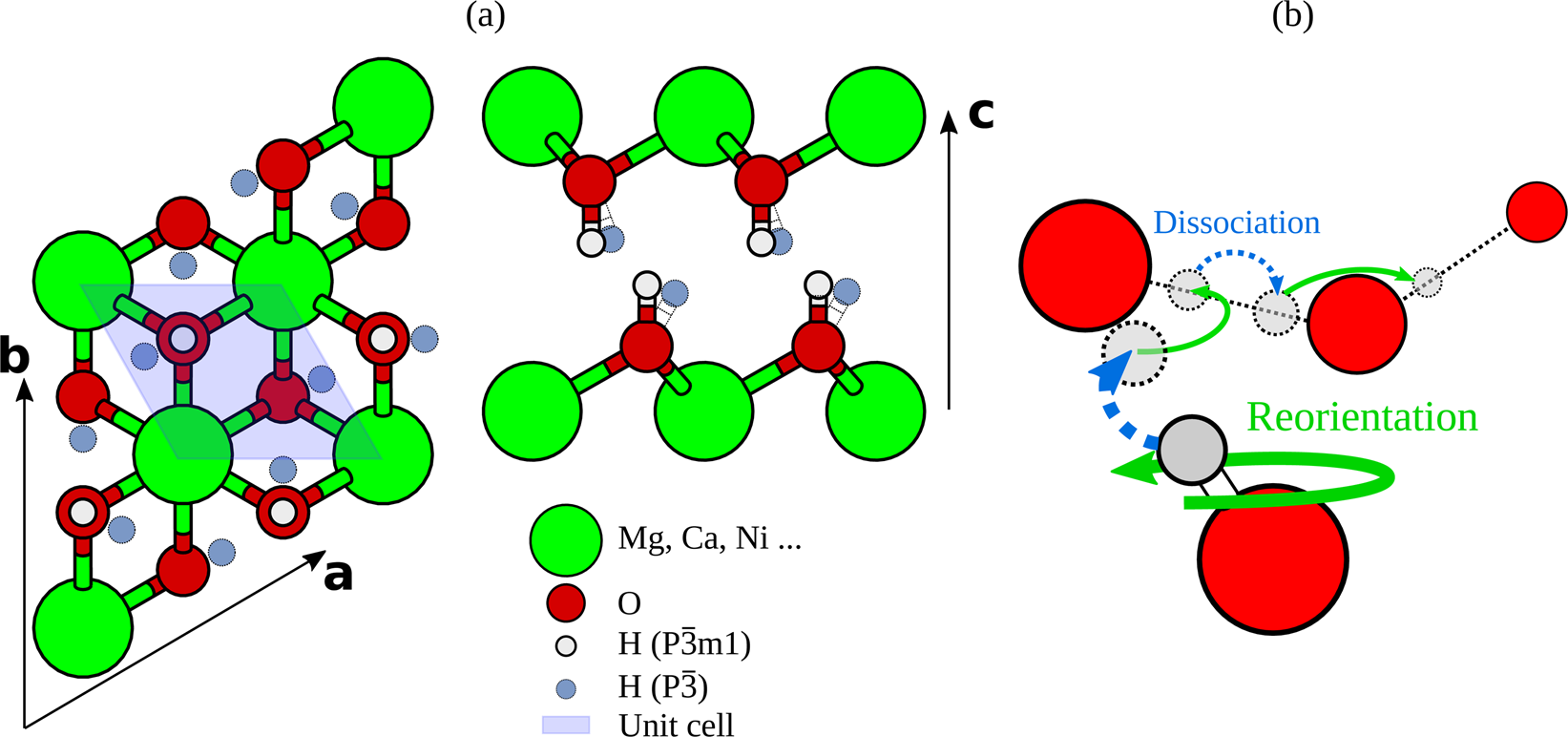
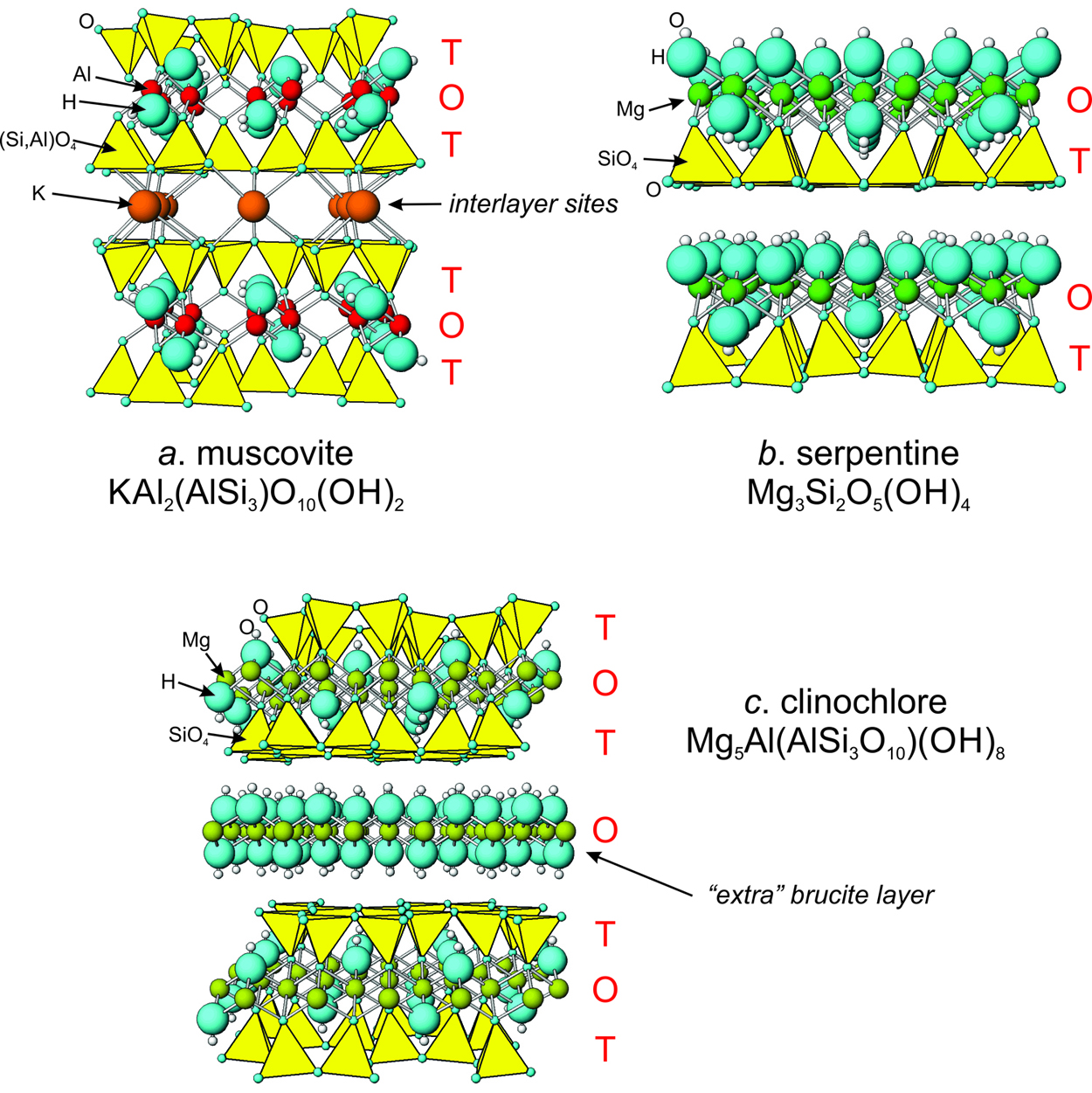
Materials | Free Full-Text | Regulation of Structure and Anion-Exchange Performance of Layered Double Hydroxide: Function of the Metal Cation Composition of a Brucite-like Layer
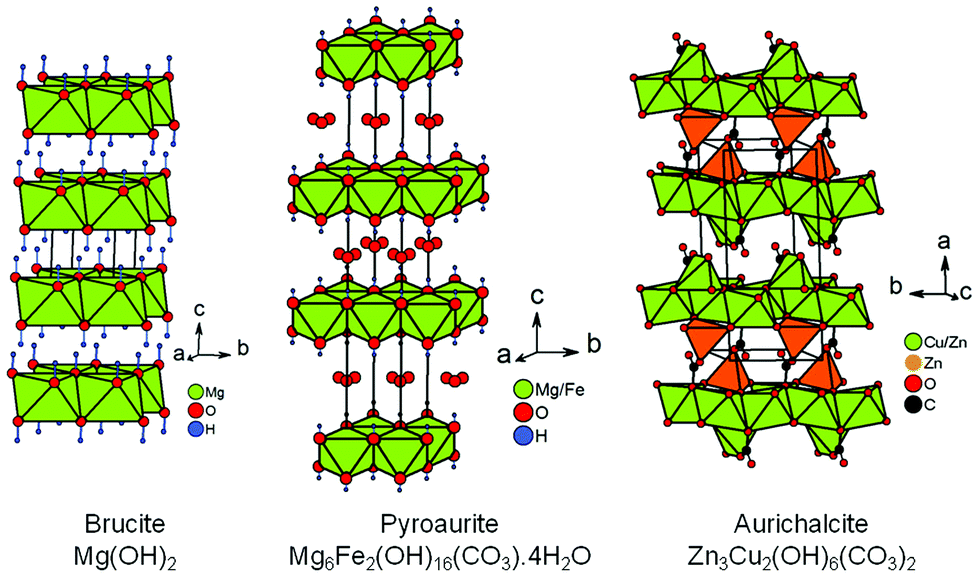
Enhanced bactericidal activity of brucite through partial copper substitution - Journal of Materials Chemistry B (RSC Publishing) DOI:10.1039/C9TB01927H

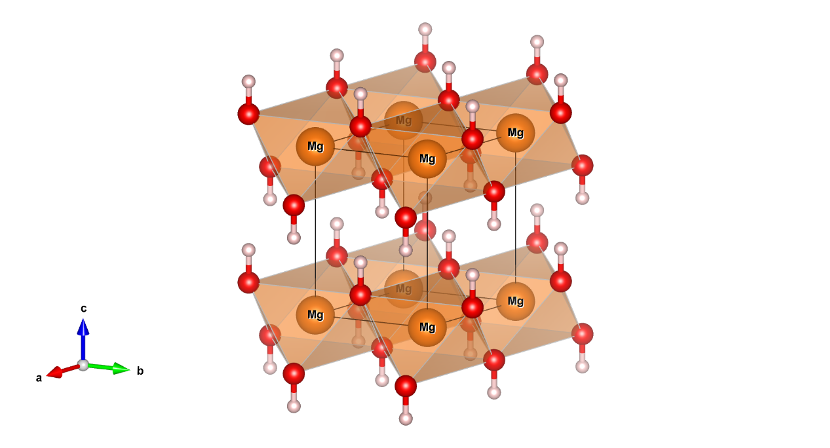
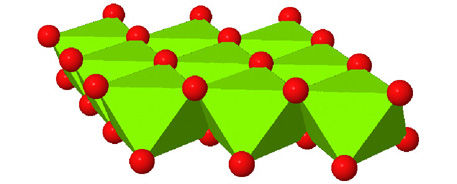
High-pressure phase of brucite stable at Earth's mantle transition zone and lower mantle conditions | PNAS

Surface Speciation of Brucite Dissolution in Aqueous Mineral Carbonation: Insights from Density-Functional Theory Simulations | The Journal of Physical Chemistry A

Binding of Nucleic Acid Components to the Serpentinite-Hosted Hydrothermal Mineral Brucite | Astrobiology
EJM - Molecular overtones and two-phonon combination bands in the near-infrared spectra of talc, brucite and lizardite

Binding of Nucleic Acid Components to the Serpentinite-Hosted Hydrothermal Mineral Brucite | Astrobiology