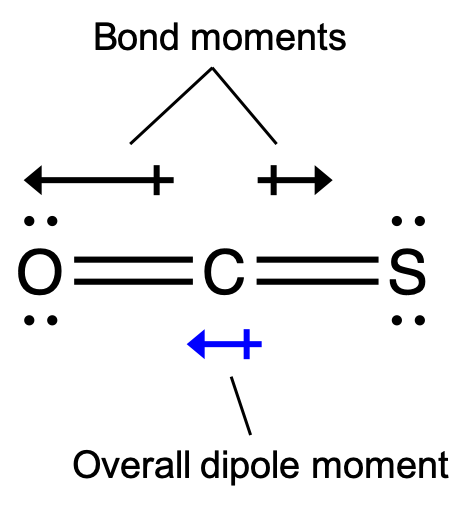
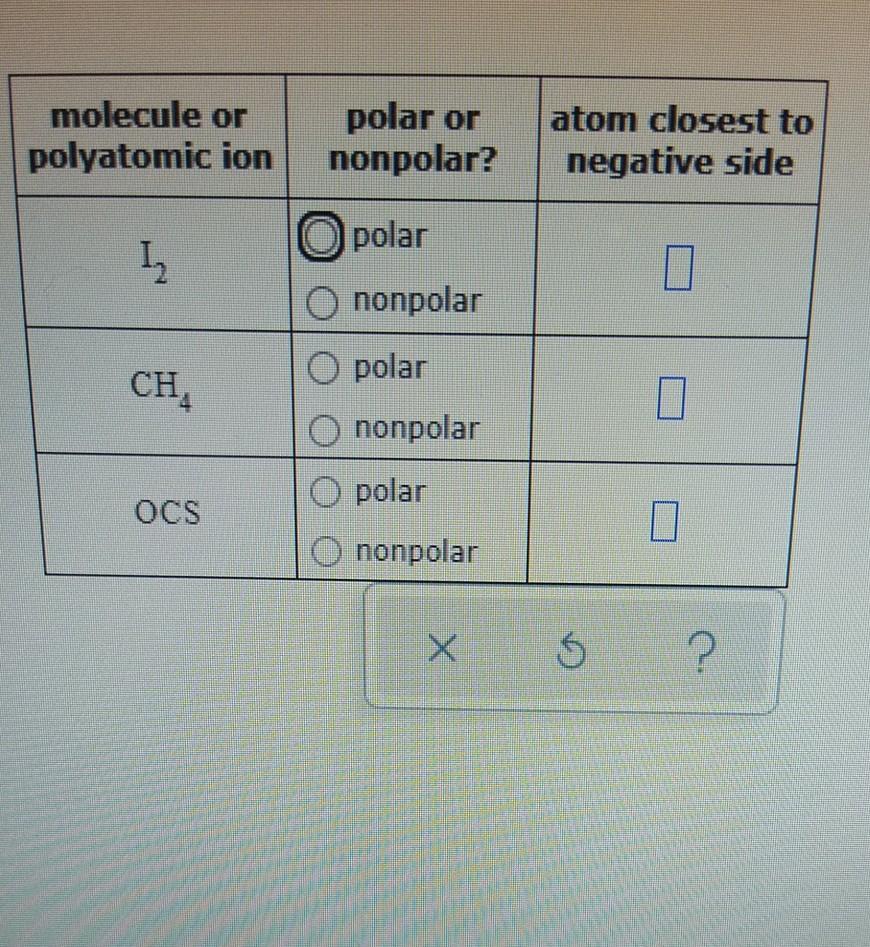
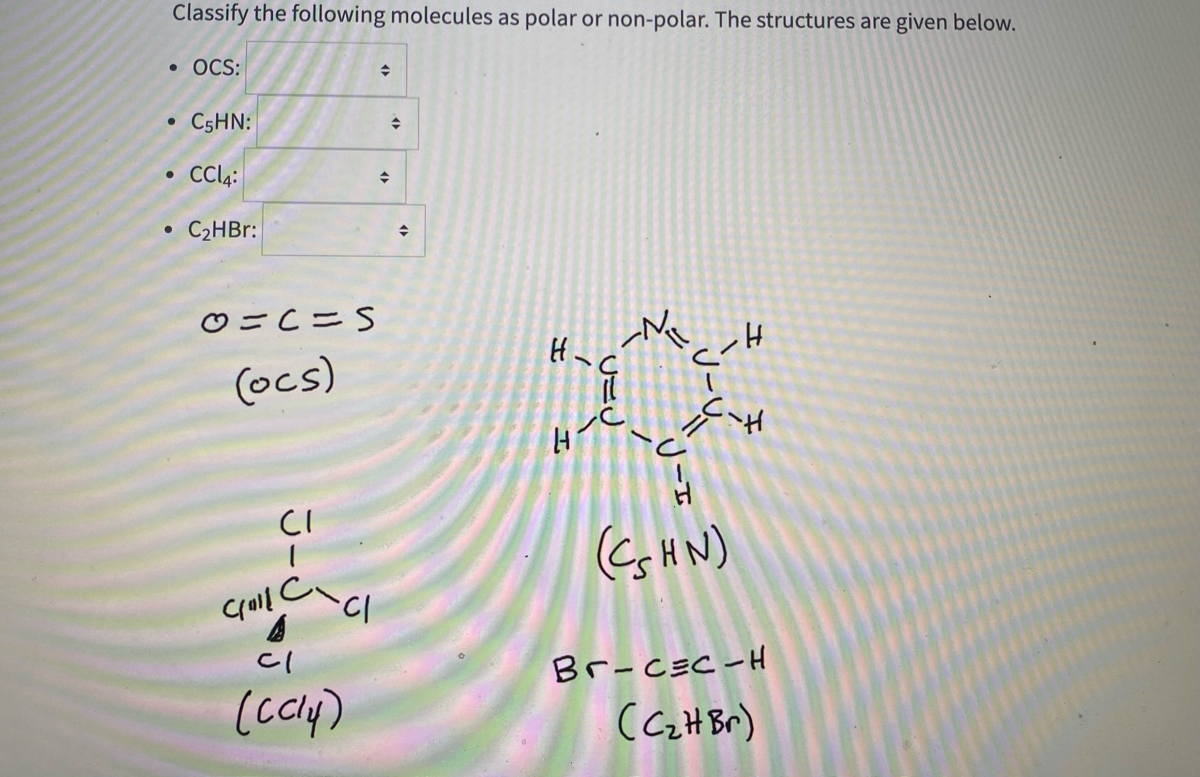
SOLVED: Text: Decide whether each molecule or polyatomic ion is polar or nonpolar: If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative

Observed structures for the known Ref. 2 nonpolar OCS dimer and for the... | Download Scientific Diagram

Observed structures for the known Ref. 2 nonpolar OCS dimer and for the... | Download Scientific Diagram

Which of the following would have the greater intermolecular forces? (a) CO_2 (b) OCS. | Homework.Study.com